Identify the incorrect statement regarding Michaelis-Menten kinetic analyses of enzyme action:
1.
the total enzyme concentration studied at each substrate concentration is fixed in analysis of enzyme kinetics.
2.
formation of enzyme-substrate complex does not appreciably decrease the concentration of substrate.
3.
Km decreases with competitive inhibition.
4.
maximal velocity is reached when the enzyme-substrate complex is equal to the total concentration of enzyme present.
If you compare the Km values for the enzymes that remove glucose from the blood in liver and brain, you expect that:
| 1. | the value will be higher for the enzyme found in brain |
| 2. | the value will be exactly ten times lower for the enzyme found in liver |
| 3. | the values for two enzymes will be same |
| 4. | the value will be lower for the enzyme found in brain |
Which of the following pairs of amino acids is only ketogenic and not glucogenic?
1. Leucine and Lysine
2. Tyrosine and Tryptophan
3. Glutamic acid and Aspartic acid
4. Alanine and Arginine
Which of the following diagrams represents the ‘micelle’ formation by amphiphilic molecules in a non polar organic solvent?
| 1. |  |
2. |  |
| 3. |  |
4. |  |
According to the International Union of Biochemistry and Molecular Biology, the enzymes belonging to EC 1 are:
| 1. | Oxidoreductases: catalyze oxidation/reduction reactions |
| 2. | Transferases: transfer a functional group (e.g. a methyl or phosphate group) |
| 3. | Hydrolases: catalyze the hydrolysis of various bonds |
| 4. | Lyases: cleave various bonds by means other than hydrolysis and oxidation |

To unlock all the explanations of 38 chapters you need to be enrolled in MasterClass Course.

To unlock all the explanations of 38 chapters you need to be enrolled in MasterClass Course.
Maltose is a disaccharide with two glucose molecules taking part in its formation. The molecular formula of maltose will be:
| 1. | C6H10O5 | 2. | C3H6O3 |
| 3. | C12H24O12 | 4. | C12H22O11 |

To unlock all the explanations of 38 chapters you need to be enrolled in MasterClass Course.

To unlock all the explanations of 38 chapters you need to be enrolled in MasterClass Course.
The number of peptide bonds in a protein molecule made of 120 amino acids will be:
| 1. | 60 | 2. | 119 |
| 3. | 120 | 4. | 240 |

To unlock all the explanations of 38 chapters you need to be enrolled in MasterClass Course.

To unlock all the explanations of 38 chapters you need to be enrolled in MasterClass Course.
Iodine-KI reagent is added to a solution or directly on a potato or other materials such as bread, crackers, or flour. A resultant blue-black color most directly indicates the presence of:
| 1. | Cellulose | 2. | Amylopectin |
| 3. | Amylose | 4. | Sucrose |
In the given graph the curve y shows the presence of an allosteric compound. This compound is acting as a/an:
1. competitive inhibitor.
2. noncompetitive inhibitor.
3. irreversible inhibitor.
4. activator.
In the given diagram showing the tertiary structure of a protein, the disulfide bond is shown by the letter:
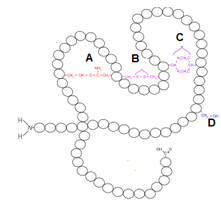
1. A
2. B
3. C
4. D

