The ratio of cations to anion in a closed pack tetrahedral is:
1. 0.714
2. 0.225
3. 0.02
4. none of these
1. 0.714
The most efficient packing of similar spheres is obtained in
1. The simple cubic system and the body centered cubic system
2. The simple cubic system and the hexagonal close packed system
3. The face centered cubic system and the hexagonal close packed system
4. The body centered cubic system and the face centered cubic system
In a compound, atoms of element Y form ccp lattice, and those of element X occupy 2/3rd of tetrahedral voids. The formula of the compound will be :-
1. X3Y4
2. X4Y3
3. X2Y3
4. X2Y
A spinel is an important class of oxides consisting of two types of metal ions with the oxide ions arranged in ccp layers. The normal spinel has 1/8th of the tetrahedral void occupied by one type of metal and one half of the octahedral voids occupied by another type of metal ions. Such a spinel is formed by Zn2+, Al3+ and O2- with Zn2+ in tetrahedral void. Give the simplest formula of the spinel.
1. ZnAl2O4
2. ZnAl2O3
3. ZnAlO
4. None of these
Close packing is maximum in the crystal lattice of:
1. simple cubic
2. face centred
3. body centred
4. none of these
Frenkel defect is noticed in:
1. AgBr
2. ZnS
3. Agl
4. All of the above
The density of KCl is 1.9893 g cm-3 and the length of a side unit cell is 6.29082 Å as determined by X-ray diffraction. The value of Avogadro’s number
calculated from this data is:-
1. 6.017 x 1023
2. 6.023 x 1022
3. 7.03 x 1023
4. 6.01 x 1019
How many octahedral and tetrahedral holes are present per unit cell in a face centred cubic arrangement of atoms?
1. 8,4
2. 1,2
3. 4,8
4. 2 ,1
TiO2 is a well-known example of:
1. Triclinic system.
2. Tetragonal system.
3. Monoclinic system.
4. None of the above.
For a solid with the following structure, the co-ordination number of the point B is:
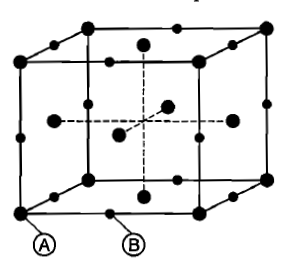
1. 3
2. 4
3. 5
4. 6
