Addition of water to alkynes occurs in acidic medium and in the presence of ions as a catalyst. Which of the following products will be formed on addition of water to but-1-yne under these conditions?
| 1. |  |
| 2. | 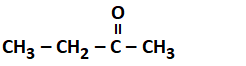 |
| 3. |  |
| 4. | 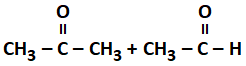 |
The reagent which does not react with both, acetone and benzaldehyde?
1. Sodium hydrogen sulphite
2. Phenyl hydrazine
3. Fehling's solution
4. Grignard reagent
The structure of ‘A’ and type of isomerism in the above reaction are respectively:
| 1. | Prop-1-en-2-ol, metamerism |
| 2. | Prop-1-en-1-ol, tautomerism |
| 3. | Prop-2-en-2-ol, geometrical isomerism |
| 4. | Prop-1-en-2-ol, tautomerism |
Compounds A and C in the following reaction are:
\(\small{\mathrm{CH}_3 \mathrm{CHO} \xrightarrow[{(ii) H_{2}O}]{(i)CH_{3}MgBr}(\mathrm{A}) \stackrel{\mathrm{H}_2 \mathrm{SO}_4}{\longrightarrow}(\mathrm{B}) \xrightarrow[]{Hydroboration \ oxidation}(C)}\)
1. Identical
2. Positional isomers
3. Functional isomers
4. Optical isomers
Which of the following compounds will give butanone on oxidation with alkaline
solution?
1. Butan-1-o1
2. Butan-2-ol
3. Both (a) and (b)
4. None of the above
Benzophenone can be obtained by .......... .
a. Benzoyl chloride + benzene
b. Benzoyl chloride + diphenyl cadmium
c. Benzoyl chloride + phenyl magnesium chloride
d. Benzene + carbon monoxide
Choose the correct option
1. (a, b)
2. (b, c)
3. (c, d)
4. (a, d)
Match the common names given in Column I with the IUPAC names given in Column II.
| Column l (Common names) |
Column ll (IUPAC names) |
| A. Cinnamaldehyde | 1. Pentanal |
| B. Acetophenone | 2. Prop-2-enal |
| C. Valeraldehyde | 3. 1-phenylethanone |
| D. Acrolein | 4. 3-Phenylprop-2-en-al |
Codes
| A | B | C | D | |
| 1. | 2 | 3 | 4 | 1 |
| 2. | 3 | 1 | 4 | 2 |
| 3. | 1 | 4 | 3 | 2 |
| 4. | 4 | 3 | 1 | 2 |
Match the reactions given in Column I with the suitable reagents given in Column II.
| Column l (Reactions) |
Column ll (Reagents) |
| A. Benzophenone → Diphenylmethane | 1. LiAlH4 |
| B. Benzaldehyde → 1-Phenylethanol | 2. DlBAL-H |
| C. Cyclohexanone → Cyclohexanol | 3. Zn(Hg)/Conc.HCl |
| D. Phenyl benzoate → Benzaldehyde | 4. CH3MgBr |
Codes
| A | B | C | D | |
| 1. | 2 | 3 | 4 | 1 |
| 2. | 3 | 4 | 1 | 2 |
| 3. | 1 | 4 | 3 | 2 |
| 4. | 4 | 3 | 2 | 1 |

