3.10 The conductivity of sodium chloride at 298 K has been determined at different concentrations and the results are given below:
Concentration/M 0.001 0.010 0.020 0.050 0.100
102 × κ/S m–1 1.237 11.85 23.15 55.53 106.74
Calculate Λm for all concentrations and draw a plot between Λm and c½. Find the value of 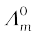 .
.
Given,
Then,
Given,
Then,
Given,
Then,
Given,
Then,
Given,
Then,
Now, we have the following data:
0.0316 | 0.1 | 0.1414 | 0.2236 | 0.3162 | |
123.7 | 118.5 | 115.8 | 111.1 | 106.74 |

Since the line interrupts at

© 2026 GoodEd Technologies Pvt. Ltd.